What Are B and T Cells Called That Have Not Yet Been Exposed to an Antigen?
Lymphocytes are responsible for the amazing specificity of adaptive immune responses. They occur in big numbers in the claret and lymph (the colorless fluid in the lymphatic vessels that connect the lymph nodes in the body to each other and to the bloodstream) and in lymphoid organs, such equally the thymus, lymph nodes, spleen, and appendix (Effigy 24-three).

Figure 24-3
Human lymphoid organs. Lymphocytes develop in the thymus and bone marrow (yellow), which are therefore called fundamental (or master) lymphoid organs. The newly formed lymphocytes migrate from these principal organs to peripheral (or secondary) lymphoid organs (more than...)
In this section, we discuss the full general properties of lymphocytes that use to both B cells and T cells. We shall encounter that each lymphocyte is committed to answer to a specific antigen and that its response during its first come across with an antigen ensures that a more rapid and effective response occurs on subsequent encounters with the aforementioned antigen. We consider how lymphocytes avoid responding to cocky antigens and how they continuously recirculate between the blood and lymphoid organs, ensuring that a lymphocyte will find its specific foreign antigen no matter where the anitgen enters the body.
Lymphocytes Are Required for Adaptive Immunity
There are about two × x12 lymphocytes in the human body, making the allowed system comparable in cell mass to the liver or brain. Despite their affluence, their central role in adaptive immunity was not demonstrated until the tardily 1950s. The crucial experiments were performed in mice and rats that were heavily irradiated to kill most of their white blood cells, including lymphocytes. This handling makes the animals unable to mount adaptive immune responses. So, by transferring various types of cells into the animals it was possible to determine which cells reversed the deficiency. Only lymphocytes restored the adaptive allowed responses of irradiated animals, indicating that lymphocytes are required for these responses (Figure 24-4).

Figure 24-four
A classic experiment showing that lymphocytes are required for adaptive immune responses to strange antigens. An of import requirement of all such jail cell-transfer experiments is that cells are transferred betwixt animals of the same inbred strain. Members (more...)
The Innate and Adaptive Immune Systems Work Together
As mentioned earlier, lymphocytes usually respond to foreign antigens simply if the innate immune arrangement is first activated. As discussed in Chapter 25, the innate allowed responses to an infection are rapid. They depend on pattern recognition receptors that recognize patterns of pathogen-associated molecules (immunostimulants) that are not present in the host organism, including microbial DNA, lipids, and polysaccharides, and proteins that form bacterial flagella. Some of these receptors are present on the surface of professional phagocytic cells such as macrophages and neutrophils, where they mediate the uptake of pathogens, which are then delivered to lysosomes for devastation. Others are secreted and bind to the surface of pathogens, marking them for destruction by either phagocytes or the complement organisation. However others are nowadays on the surface of various types of host cells and actuate intracellular signaling pathways in response to the binding of pathogen-associated immunostimulants; this leads to the production of extracellular betoken molecules that promote inflammation and help activate adaptive immune responses.
Some cells of the innate immune system straight nowadays microbial antigens to T cells to initiate an adaptive immune response. The cells that do this most efficiently are called dendritic cells, which are present in most vertebrate tissues. They recognize and phagocytose invading microbes or their products at a site of infection and and then migrate with their prey to a nearby peripheral lymphoid organ. There they act as antigen-presenting cells, which directly activate T cells to respond to the microbial antigens. Once activated, some of the T cells so migrate to the site of infection, where they aid other phagocytic cells, mainly macrophages, destroy the microbes (Figure 24-5). Other activated T cells remain in the lymphoid organ and help B cells respond to the microbial antigens. The activated B cells secrete antibodies that circulate in the body and coat the microbes, targeting them for efficient phagocytosis.

Figure 24-5
One mode in which the innate immune system helps activate the adaptive immune arrangement. Specialized phagocytic cells of the innate immune organisation, including macrophages (non shown) and dendritic cells ingest invading microbes or their products at the site (more than...)
Thus, innate allowed responses are activated mainly at sites of infection, whereas adaptive immune responses are activated in peripheral lymphoid organs. The two types of responses piece of work together to eliminate invading pathogens.
B Lymphocytes Develop in the Bone Marrow; T Lymphocytes Develop in the Thymus
T cells and B cells derive their names from the organs in which they develop. T cells develop in the thymus, and B cells, in mammals, develop in the bone marrow in adults or the liver in fetuses.
Despite their unlike origins, both T and B cells develop from the aforementioned pluripotent hemopoietic stem cells, which requite rise to all of the blood cells, including red blood cells, white blood cells, and platelets. These stem cells (discussed in Chapter 22) are located primarily in hemopoietic tissues—mainly the liver in fetuses and the bone marrow in adults. T cells develop in the thymus from precursor cells that migrate in that location from the hemopoietic tissues via the blood. In well-nigh mammals, including humans and mice, B cells develop from stalk cells in the hemopoietic tissues themselves (Figure 24-6). Because they are sites where lymphocytes develop from forerunner cells, the thymus and hemopoietic tissues are referred to as central (primary) lymphoid organs (see Figure 24-3).

Figure 24-6
The evolution and activation of T and B cells. The key lymphoid organs, where lymphocytes develop from forerunner cells, are labeled in yellow boxes. Lymphocytes respond to antigen in peripheral lymphoid organs, such as lymph nodes or spleen.
As nosotros hash out later on, most lymphocytes die in the central lymphoid organ soon after they develop, without ever functioning. Others, however, mature and migrate via the claret to the peripheral (secondary) lymphoid organs—mainly, the lymph nodes, spleen, and epithelium-associated lymphoid tissues in the gastrointestinal tract, respiratory tract, and skin (see Figure 24-3). Every bit mentioned earlier, it is in the peripheral lymphoid organs that T cells and B cells react with foreign antigens (see Figure 24-6).
T and B cells become morphologically distinguishable from each other only afterwards they have been activated by antigen. Nonactivated T and B cells look very similar, even in an electron microscope. Both are small-scale, only marginally bigger than red claret cells, and comprise piffling cytoplasm (Figure 24-7A). Both are activated past antigen to proliferate and mature into effector cells. Effector B cells secrete antibodies. In their most mature grade, chosen plasma cells, they are filled with an extensive rough endoplasmic reticulum (Figure 24-7B). In contrast, effector T cells (Figure 24-7C) contain very petty endoplasmic reticulum and do not secrete antibodies.

Figure 24-seven
Electron micrographs of nonactivated and activated lymphocytes. (A) A resting lymphocyte, which could be a T cell or a B cell, every bit these cells are difficult to distinguish morphologically until they take been activated to become effector cells. (B) An (more than...)
There are two main classes of T cells—cytotoxic T cells and helper T cells. Cytotoxic T cells kill infected cells, whereas helper T cells aid activate macrophages, B cells, and cytotoxic T cells. Effector helper T cells secrete a variety of point proteins called cytokines, which act equally local mediators. They also display a variety of costimulatory proteins on their surface. By means of these cytokines and membrane-spring costimulatory proteins, they can influence the behavior of the diverse cell types they help. Effector cytotoxic T cells kill infected target cells too by means of proteins that they either secrete or brandish on their surface. Thus, whereas B cells can deed over long distances past secreting antibodies that are distributed by the bloodstream, T cells can migrate to distant sites, but there they act just locally on neighboring cells.
The Adaptive Immune System Works by Clonal Selection
The most remarkable characteristic of the adaptive immune system is that it can answer to millions of dissimilar strange antigens in a highly specific way. B cells, for case, make antibodies that react specifically with the antigen that induced their product. How do B cells produce such a diversity of specific antibodies? The answer began to sally in the 1950s with the formulation of the clonal selection theory. According to this theory, an beast first randomly generates a vast diversity of lymphocytes, and so those lymphocytes that can react against the foreign antigens that the fauna actually encounters are specifically selected for activeness. As each lymphocyte develops in a central lymphoid organ, it becomes committed to react with a item antigen earlier always being exposed to the antigen. It expresses this commitment in the form of cell-surface receptor proteins that specifically fit the antigen. When a lymphocyte encounters its antigen in a peripheral lymphoid organ, the bounden of the antigen to the receptors activates the lymphocyte, causing information technology both to proliferate and to differentiate into an effector jail cell. An antigen therefore selectively stimulates those cells that express complementary antigen-specific receptors and are thus already committed to reply to it. This organisation is what makes adaptive immune responses antigen-specific.
The term "clonal" in clonal selection theory derives from the postulate that the adaptive immune arrangement is composed of millions of different families, or clones, of lymphocytes, each consisting of T or B cells descended from a common antecedent. Each ancestral prison cell was already committed to make one item antigen-specific receptor protein, and and so all cells in a clone have the same antigen specificity (Effigy 24-8). According to the clonal selection theory, then, the immune system functions on the "ready-made" principle rather than the "made-to-club" ane.

Figure 24-8
The clonal choice theory. An antigen activates only those lymphocyte clones (represented here by single cells) that are already committed to reply to it. A cell committed to respond to a item antigen displays cell-surface receptors that specifically (more...)
There is compelling evidence to support the main tenets of the clonal selection theory. For example, when lymphocytes from an animal that has not been immunized are incubated in a test tube with a number of radioactively labeled antigens, only a very small proportion (less than 0.01%) bind each antigen, suggesting that only a few cells are committed to respond to these antigens. Moreover, when ane antigen is made so highly radioactive that it kills any prison cell that it binds to, the remaining lymphocytes can no longer produce an immune response to that item antigen, even though they can all the same respond normally to other antigens. Thus, the committed lymphocytes must have receptors on their surface that specifically bind that antigen. Although almost experiments of this kind have involved B cells and antibiotic responses, other experiments indicate that T cells, like B cells, operate past clonal selection.
How tin can the adaptive immune system produce lymphocytes that collectively brandish such an enormous diversity of receptors, including ones that recognize synthetic molecules that never occur in nature? Nosotros shall see later that the antigen-specific receptors on both T and B cells are encoded past genes that are assembled from a series of gene segments by a unique grade of genetic recombination that occurs early in a lymphocyte's development, before it has encountered antigen. This assembly process generates the enormous variety of receptors and lymphocytes, thereby enabling the immune system to respond to an almost unlimited diversity of antigens.
Nigh Antigens Actuate Many Dissimilar Lymphocyte Clones
Nigh large molecules, including most all proteins and many polysaccharides, tin serve every bit antigens. Those parts of an antigen that combine with the antigen-binding site on either an antibiotic molecule or a lymphocyte receptor are chosen antigenic determinants (or epitopes). Most antigens have a variety of antigenic determinants that can stimulate the production of antibodies, specific T prison cell responses, or both. Some determinants of an antigen produce a greater response than others, so that the reaction to them may dominate the overall response. Such determinants are said to exist immunodominant.
The diversity of lymphocytes is such that even a single antigenic determinant is likely to activate many clones, each of which produces an antigen-bounden site with its own characteristic affinity for the determinant. Even a relatively simple structure, like the dinitrophenyl (DNP) group in Effigy 24-9, can be "looked at" in many ways. When it is coupled to a protein, as shown in the figure, information technology usually stimulates the production of hundreds of species of anti-DNP antibodies, each made by a different B cell clone. Such responses are said to be polyclonal. When only a few clones are activated, the response is said to be oligoclonal; and when the response involves only a single B or T cell clone, it is said to be monoclonal. Monoclonal antibodies are widely used as tools in biology and medicine, but they accept to be produced in a special way (see Figure 8-6), every bit the responses to most antigens are polyclonal.

Figure 24-9
The dinitrophenyl (DNP) grouping. Although it is too modest to induce an immune response on its own, when it is coupled covalently to a lysine side chain on a protein, equally illustrated, DNP stimulates the product of hundreds of different species of antibodies (more...)
Immunological Memory Is Due to Both Clonal Expansion and Lymphocyte Differentiation
The adaptive immune organisation, like the nervous system, can remember prior experiences. This is why nosotros develop lifelong immunity to many mutual infectious diseases after our initial exposure to the pathogen, and information technology is why vaccination works. The aforementioned phenomenon tin can be demonstrated in experimental animals. If an animate being is immunized once with antigen A, an immune response (either antibiotic or cell-mediated) appears afterwards several days, rises rapidly and exponentially, and then, more gradually, declines. This is the characteristic class of a master allowed response, occurring on an creature's first exposure to an antigen. If, after some weeks, months, or even years have elapsed, the creature is reinjected with antigen A, it volition usually produce a secondary immune response that is very different from the primary response: the lag menstruum is shorter, and the response is greater. These differences indicate that the animal has "remembered" its first exposure to antigen A. If the animal is given a different antigen (for example, antigen B) instead of a second injection of antigen A, the response is typical of a principal, and not a secondary, immune response. The secondary response must therefore reflect antigen-specific immunological memory for antigen A (Figure 24-10).
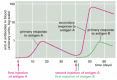
Figure 24-10
Master and secondary antibody responses. The secondary response induced by a 2nd exposure to antigen A is faster and greater than the primary response and is specific for A, indicating that the adaptive allowed system has specifically remembered encountering (more...)
The clonal selection theory provides a useful conceptual framework for understanding the cellular footing of immunological memory. In an developed beast, the peripheral lymphoid organs contain a mixture of cells in at to the lowest degree three stages of maturation: naïve cells, effector cells and memory cells. When naïve cells encounter antigen for the showtime time, some of them are stimulated to proliferate and differentiate into effector cells, which are actively engaged in making a response (effector B cells secrete antibody, while effector T cells kill infected cells or help other cells fight the infection). Instead of becoming effector cells, some naïve cells are stimulated to multiply and differentiate into retentiveness cells—cells that are not themselves engaged in a response but are more easily and more than speedily induced to get effector cells by a later encounter with the same antigen. Memory cells, like naïve cells, give rise to either effector cells or more memory cells (Effigy 24-xi).
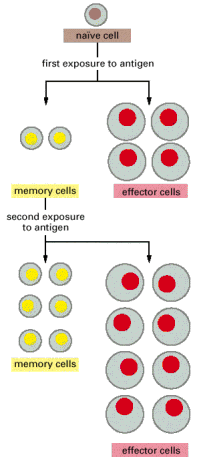
Figure 24-11
A model for the cellular basis of immunological memory. When naïve lymphocytes are stimulated by their specific antigen, they proliferate and differentiate. Most become effector cells which part and so die, while others become long-lived (more...)
Thus, immunological memory is generated during the primary response in part considering the proliferation of antigen-stimulated naïve cells creates many memory cells—a process known every bit clonal expansion—and in part because retention cells are able to answer more sensitively and apace to the same antigen than do naïve cells. And, unlike nigh effector cells, which dice within days or weeks, memory cells can live for the lifetime of the animate being, thereby providing lifelong immunological memory.
Caused Immunological Tolerance Ensures That Cocky Antigens Are Not Attacked
As discussed in Chapter 25, cells of the innate immune organization recognize molecules on the surface of pathogens that are not found in the host. The adaptive immune system has a far more difficult recognition task: information technology must be able to respond specifically to an nearly unlimited number of foreign macromolecules, while avoiding responding to the large number of molecules fabricated by the host organism itself. How does it practise it? For one matter, self molecules do not induce the innate immune reactions that are required to activate adaptive immune responses. Simply even when an infection triggers innate reactions, cocky molecules still exercise not normally induce adaptive allowed responses. Why not?
One answer is that the adaptive immune system "learns" not to respond to self antigens. Transplantation experiments provide one line of show for this learning process. When tissues are transplanted from one individual to another, equally long every bit the two individuals are not identical twins, the immune system of the recipient usually recognizes the donor cells equally foreign and destroys them. (For reasons we hash out later on, the foreign antigens on the donor cells are and then powerful that they can stimulate adaptive allowed responses in the absence of infection or an adjuvant.) If, however, cells from one strain of mouse are introduced into a neonatal mouse of some other strain, some of these cells survive for most of the recipient fauna's life, and the recipient will now have a graft from the original donor, even though information technology rejects "third-political party" grafts. Apparently, nonself antigens tin, in some circumstances, induce the immune arrangement to get specifically unresponsive to them. This antigen-specific unresponsiveness to foreign antigens is known as acquired immunological tolerance (Effigy 24-12).

Figure 24-12
Immunological tolerance. The peel graft seen here, transplanted from an adult brown mouse to an adult white mouse, has survived for many weeks but because the white mouse, at the fourth dimension of its nascence, received an injection of cells from the brown mouse (more...)
The unresponsiveness of an animal's adaptive immune system to its own macromolecules (natural immunological tolerance) is acquired in the same way. Normal mice, for example, cannot make an immune response confronting one of their own protein components of the complement system called C5 (discussed in Chapter 25). Mutant mice, however, that lack the cistron encoding C5 (but are otherwise genetically identical to the normal mice) can make a stiff allowed response to this blood poly peptide when immunized with information technology. Natural immunological tolerance for a detail cocky molecule persists only for every bit long as the molecule remains present in the body. If a self molecule such every bit C5 is removed, an brute gains the ability to respond to it afterwards a few weeks or months. Thus, the allowed arrangement is genetically capable of responding to self molecules only learns non to do and so.
The learning procedure that leads to cocky-tolerance can involve killing the self-reactive lymphocytes (clonal deletion), functionally inactivating them (clonal anergy or inactivation), stimulating the cells to produce modified receptors that no longer recognize the self antigen (receptor editing), or the suppression of self-reactive lymphocytes by a special type of regulatory T cell. The process begins in the cardinal lymphoid organs when newly formed self-reactive lymphocytes first encounter their self antigen. Instead of being activated by binding antigen, the young lymphocytes are induced to either alter their receptors or die past apoptosis. Lymphocytes that could potentially respond to self antigens that are not present in the central lymphoid organs often die or are either inactivated or suppressed after they have matured and migrated to peripheral lymphoid organs.
Why does the binding of cocky antigen lead to tolerance rather than activation? Equally we discuss after, for a lymphocyte to be activated in a peripheral lymphoid organ, it must not simply bind its antigen but must besides receive a costimulatory signal. The latter indicate is provided by a helper T prison cell in the case of a B lymphocyte and past an antigen-presenting jail cell in the case of a T lymphocyte. The production of costimulatory signals usually depends on exposure to pathogens, and so a self-reactive lymphocyte ordinarily encounters its antigen in the absence of such signals. Without a costimulatory betoken, an antigen tends to impale or inactivate a lymphocyte rather than activate information technology (Figure 24-13).

Figure 24-xiii
Induction of immunological tolerance to self antigens in central and peripheral lymphoid organs. When a self-reactive young lymphocyte binds its self antigen in the fundamental lymphoid organ where the prison cell is produced, it may be induced to alter the receptor (more...)
Tolerance to self antigens sometimes breaks downwards, causing T or B cells (or both) to react against the organism's own tissue antigens. Myasthenia gravis is an instance of such an autoimmune disease. Affected individuals brand antibodies against the acetylcholine receptors on their own skeletal muscle cells. These antibodies interfere with the normal functioning of the receptors and then that the patients become weak and may die considering they cannot exhale. The mechanisms responsible for the breakdown of tolerance to cocky antigens in autoimmune diseases are unknown. Information technology is thought, yet, that activation of the innate allowed system by infection may help trigger certain anti-self responses in genetically susceptible individuals.
Lymphocytes Continuously Circulate Through Peripheral Lymphoid Organs
Pathogens generally enter the body through an epithelial surface, usually through the pare, gut, or respiratory tract. How practise the microbial antigens travel from these entry points to a peripheral lymphoid organ, such every bit a lymph node or the spleen, where lymphocytes are activated (come across Effigy 24-half dozen)? The route and destination depend on the site of entry. Antigens that enter through the skin or respiratory tract are carried via the lymph to local lymph nodes; those that enter through the gut stop upward in gut-associated peripheral lymphoid organs such as Peyer's patches; and those that enter the blood are filtered out in the spleen. In almost cases, dendritic cells carry the antigen from the site of infection to the peripheral lymphoid organ, where they become antigen-presenting cells (see Figure 24-v), specialized for activating T cells (as nosotros discuss later).
But the lymphocytes that can recognize a detail microbial antigen in a peripheral lymph organ are just a tiny fraction of the full lymphocyte population. How do these rare cells find an antigen-presenting cell displaying their antigen? The reply is that they continuously circulate betwixt the lymph and blood until they encounter their antigen. In a lymph node, for example, lymphocytes continually exit the bloodstream by squeezing out between specialized endothelial cells lining small veins chosen postcapillary venules. Afterwards percolating through the node, they accumulate in small lymphatic vessels that leave the node and connect with other lymphatic vessels that pass through other lymph nodes downstream (run into Figure 24-3). Passing into larger and larger vessels, the lymphocytes eventually enter the main lymphatic vessel (the thoracic duct), which carries them back into the blood (Figure 24-14). This continuous recirculation between the claret and lymph ends only if a lymphocyte encounters its specific antigen (and a costimulatory signal) on the surface of an antigen-presenting cell in a peripheral lymphoid organ. Now the lymphocyte is retained in the peripheral lymphoid organ, where it proliferates and differentiates into effector cells. Some of the effector T cells then get out the organ via the lymph and migrate through the blood to the site of infection (come across Figure 24-five).

Figure 24-14
The path followed by lymphocytes as they continuously circulate between the lymph and claret. The apportionment through a lymph node is shown here. Microbial antigens are carried into the lymph node by dendritic cells, which enter via afferent lymphatic (more...)
Lymphocyte recirculation depends on specific interactions between the lymphocyte prison cell surface and the surface of the specialized endothelial cells lining the postcapillary venules in the peripheral lymphoid organs. Many cell types in the claret come into contact with these endothelial cells, but just lymphocytes adhere and so migrate out of the bloodstream. The lymphocytes initially adhere to the endothelial cells via homing receptors that bind to specific ligands (often chosen counterreceptors) on the endothelial cell surface. Lymphocyte migration into lymph nodes, for example, depends on a homing receptor poly peptide called L-selectin, a member of the selectin family of cell-surface lectins discussed in Chapter xix. This protein binds to specific sugar groups on a counterreceptor that is expressed exclusively on the surface of the specialized endothelial cells in lymph nodes, causing the lymphocytes to adhere weakly to the endothelial cells and to gyre slowly along their surface. The rolling continues until another, much stronger adhesion organization is called into play by chemo-attractant proteins (chosen chemokines; see below) secreted by endothelial cells. This strong adhesion is mediated past members of the integrin family of cell adhesion molecules (discussed in Chapter 19), which become activated on the lymphocyte surface. Now the lymphocytes terminate rolling and crawl out of the blood vessel into the lymph node (Figure 24-fifteen).

Figure 24-15
Migration of a lymphocyte out of the bloodstream into a lymph node. A circulating lymphocyte adheres weakly to the surface of the specialized endothelial cells lining a postcapillary venule in a lymph node. This initial adhesion is mediated by 50-selectin (more than...)
Chemokines are small, secreted, positively charged proteins that have a central role in guiding the migrations of diverse types of white blood cells. They are all structurally related and bind to the surface of endothelial cells, and to negatively charged proteoglycans of the extracellular matrix in organs. By bounden to G-protein-linked receptors (discussed in Chapter 15) on the surface of specific blood cells, chemokines concenter these cells from the bloodstream into an organ, guide them to specific locations within the organ, and and then aid stop migration. (The AIDS virus (HIV) besides binds to chemokine receptors, which allows the virus to infect white blood cells.) T and B cells initially enter the same region of a lymph node but are then attracted by dissimilar chemokines to separate regions of the node—T cells to the paracortex and B cells to lymphoid follicles (Figure 24-xvi). Unless they encounter their antigen, both types of cells shortly leave the lymph node via lymphatic vessels. If they encounter their antigen, nevertheless, they remain in the node, proliferate, and differentiate into either effector cells or memory cells. Most of the effector cells leave the node, expressing dissimilar chemokine receptors that assist guide them to their new destinations—T cells to sites of infection and B cells to the bone marrow.
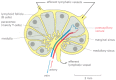
Figure 24-xvi
A simplified cartoon of a human lymph node. B cells are primarily clustered in structures called lymphoid follicles, whereas T cells are found mainly in the paracortex. Both types of lymphocytes are attracted past chemokines to enter the lymph node from (more...)
Summary
Innate immune responses are triggered at sites of infection by microbe-specific molecules associated with invading pathogens. In improver to fighting infection straight, these responses help activate adaptive allowed responses in peripheral lymphoid organs. Unlike innate allowed responses, adaptive responses provide specific and long-lasting protection against the particular pathogen that induced them.
The adaptive immune organisation is equanimous of millions of lymphocyte clones, with the cells in each clone sharing a unique jail cell-surface receptor that enables them to bind a particular antigen. The binding of antigen to these receptors, all the same, is normally not sufficient to stimulate a lymphocyte to proliferate and differentiate into an effector cell that can assist eliminate the pathogen. Costimulatory signals provided by another specialized cell in a peripheral lymphoid organ are besides required. Helper T cells provide such signals for B cells, while antigen-presenting dendritic cells commonly provide them for T cells. Effector B cells secrete antibodies, which tin act over long distances to help eliminate extracellular pathogens and their toxins. Effector T cells, by dissimilarity, act locally at sites of infection to either impale infected host cells or help other cells to eliminate pathogens. As function of the adaptive allowed response, some lymphocytes proliferate and differentiate into memory cells, which are able to respond faster and more efficiently the next time the same pathogen invades. Lymphocytes that would react against self molecules are either induced to change their receptors, induced to kill themselves, inactivated, or suppressed, so that the adaptive immune system ordinarily reacts just against strange antigens. Both B and T cells broadcast continuously between the claret and lymph. Only if they encounter their specific foreign antigen in a peripheral lymphoid organ do they stop migrating, proliferate, and differentiate into effector cells or memory cells.

Source: https://www.ncbi.nlm.nih.gov/books/NBK26921/
0 Response to "What Are B and T Cells Called That Have Not Yet Been Exposed to an Antigen?"
Post a Comment